
CAFFEINE PHENACETIN BUTALBITAL 10664 PANABARB PAN. All patients who have filled prescriptions of Hydrocodone Bitartrate and Acetaninophen manufactured by Qualitest, are asked to double check the identity of their tablets. CAFFEINE 09663 PALGESIC PAN AMERICAN LABS CA 3 PHENACETIN ACETAMINOPHEN. RECOMMENDATION : Qualitest is notifying all customers who may have received affected product and arranging for the return of any affected product. Tablet descriptions and photographs of tablets for these two products are provided in the firm's press release. Lot numbers can be found on the side of the manufacturer’s bottle. These lots were distributed between Novemand Apto wholesale and retail pharmacies nationwide (including Puerto Rico). Hydrocodone Bitartrate and Acetaminophen Tablets, USP 7.5mg / 500mg, NDC 0603-3882-32, 1000 count, Lot Numbers C0390909A, C0400909A, C0410909A, C0590909B oxycodone oral brand names and other generic formulations include: Dazidox Oral, Endocodone Oral, ETH-Oxydose Oral, Oxaydo Oral, Oxecta Oral, Ox圜ontin Oral, Oxyfast Oral, OxyIR Oral, Percolone Oral, Roxicodone Intensol Oral, Roxicodone Oral. 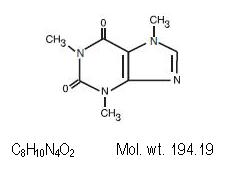
Patients who were receiving hydrocodone for chronic pain might experience worsening pain and withdrawal symptoms as a result of this substitution.īACKGROUND : The recall includes the following products: Butalbital, acetaminophen and caffeine capsules, USP containing butalbital 50 mg (Warning: May be habit-forming), acetaminophen 300 mg and caffeine 40 mg, are supplied as White to off-white powder filled in a hard gelatin capsule with a Light Blue opaque cap and body, imprinted with 'A3l' on cap and body in black ink. Side effects due to caffeine are less likely, due to the small amounts in this formulation, however, those individuals with a sensitivity to caffeine may experience symptoms such as tremors, irritability, and difficulty sleeping. Store at 20 to 25C (68 to 77F) see USP Controlled Room Temperature. 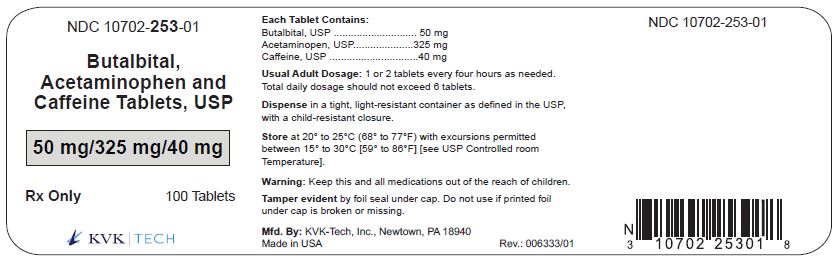
Additionally, patients with an allergy to butalbital could experience a hypersensitivity reaction. Butalbital, Acetaminophen, and Caffeine Tablets USP, 50 mg/325mg/40 mg are light-blue, speckled, round uncoated tablets, debossed '1695' on one side and 'LCI' on the other side and are supplied in bottles of 100 (NDC 0527-1695-01) and bottles of 500 (NDC 0527-1695-05). Unintentional administration of butalbital could result in symptoms such as sedation, lightheadedness, dizziness, and nausea. See also Warning section.This combination medication is used to treat tension headaches. ISSUE : Qualitest and FDA is notifying pharmacists and patients of a recall to the retail level of specific lots of Butalbital, Acetaminophen, and Caffeine Tablets and Hydrocodone Bitartrate and Acetaminophen Tablets due to possibility that the recalled bottles may contain incorrect tablets, and patients may unintentionally take butalbital and caffeine instead of hydrocodone. Butal-ASA-Caff-Cod Capsule - Uses, Side Effects, and More.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
